Intermolecular forces worksheets help students understand why substances behave differently even when their molecules may look simple on paper. In chemistry, learners often study bonds inside a molecule first, but they also need to understand the attractions between molecules. These forces explain why water has a high boiling point, why some substances evaporate quickly, and why certain molecules dissolve better than others. With structured practice, students can move from memorizing force names to explaining real physical properties with confidence.
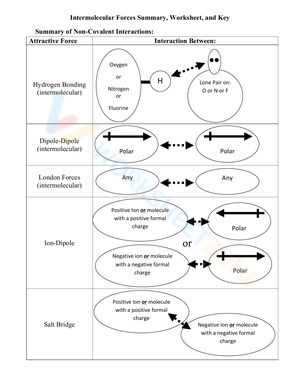
A strong worksheet on intermolecular forces usually begins with the main types of attractions: London dispersion forces, dipole-dipole interactions, and hydrogen bonding. Students may compare molecules, identify polarity, predict the strongest force present, or rank substances by boiling point. Before studying these attractions in depth, it is helpful for learners to review how atoms connect inside compounds. Teachers can pair this topic with ionic and covalent bonding exercises so students understand the difference between chemical bonds and intermolecular attractions.
Intermolecular forces worksheets are especially useful because this topic combines several chemistry skills at once. Students need to read molecular formulas, understand electronegativity, recognize molecular shape, and connect structure to properties such as melting point, boiling point, viscosity, and solubility. A worksheet might ask students to compare water, methane, ammonia, and carbon dioxide, then explain why one substance has stronger attractions than another. This kind of practice helps learners see chemistry as a system of patterns rather than a list of unrelated facts.
These worksheets can also strengthen students’ reasoning with the periodic table. Electronegativity, atomic size, and molecular composition all influence how molecules interact. Teachers who want to make this background knowledge more engaging can use periodic table activity ideas before moving into intermolecular force analysis. When students understand periodic trends, they are better prepared to explain why polar molecules behave differently from nonpolar molecules and why hydrogen bonding creates unusually strong attractions in certain substances.
Worksheetzone’s intermolecular forces worksheets are useful for high school chemistry, honors chemistry, AP Chemistry review, and introductory college practice. Teachers can use them as guided notes, homework, quiz review, group practice, lab preparation, or exit tickets. Students benefit from repeated exposure to molecule comparisons, property predictions, and explanation-based questions. With consistent practice, intermolecular forces become easier to identify, easier to explain, and more meaningful in real chemistry applications.
Frequently Asked Questions
Question 1: What topics do intermolecular forces worksheets cover?
These worksheets usually cover London dispersion forces, dipole-dipole interactions, hydrogen bonding, molecular polarity, electronegativity, boiling point trends, melting point trends, and solubility. Some worksheets also ask students to compare molecules and explain how intermolecular attractions affect physical properties.
Question 2: What grade levels are intermolecular forces worksheets best for?
Intermolecular forces worksheets are most useful for high school chemistry students, especially grades 10 through 12. They can also support honors chemistry, AP Chemistry, and introductory college chemistry courses. Students should already have some background in covalent bonding, polarity, and molecular structure before starting this topic.
Question 3: Why do students often struggle with intermolecular forces?
Students often confuse intermolecular forces with chemical bonds. Chemical bonds hold atoms together inside a molecule, while intermolecular forces attract separate molecules to one another. Learners may also struggle to connect polarity, molecular shape, and physical properties. Focused worksheet practice helps make these differences clearer.
Question 4: How can teachers use these worksheets in class?
Teachers can use intermolecular forces worksheets after lessons on bonding, polarity, or molecular geometry. They work well as guided practice, homework, partner activities, review pages, quiz preparation, or lab follow-ups. They are especially helpful when students need to explain boiling point, solubility, or phase behavior using evidence from molecular structure.