1 / 2
0
Views
0
Downloads

0.0
0
Save
0 Likes
Periodic Table Practice Worksheet
0 Views
0 Downloads
Paste this activity's link or code into your existing LMS (Google Classroom, Canvas, Teams, Schoology, Moodle, etc.).
Students can open and work on the activity right away, with no student login required.
You'll still be able to track student progress and results from your teacher account.
Information
Description
What It Is:
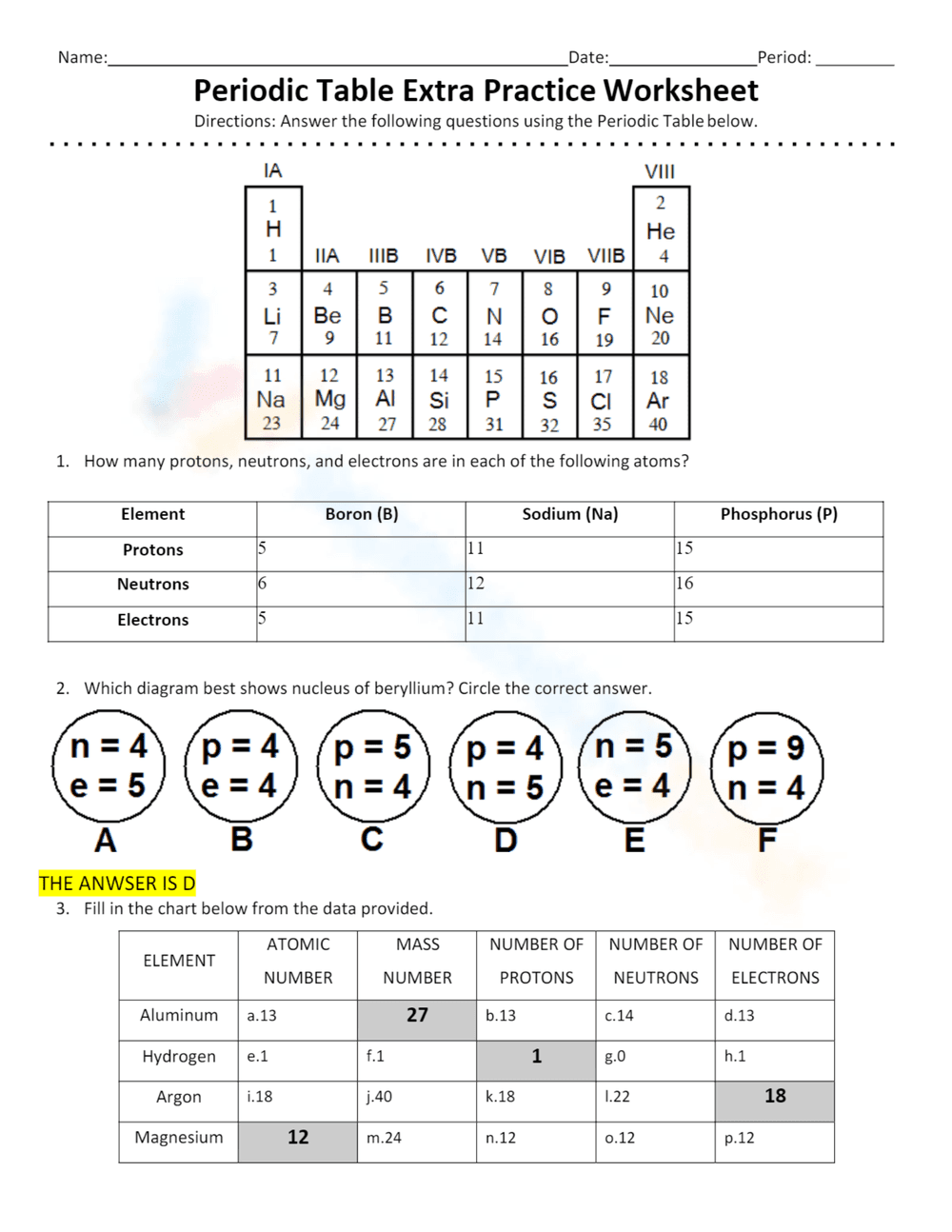
This is a Periodic Table extra practice worksheet. It includes a simplified periodic table at the top. Question 1 asks students to determine the number of protons, neutrons, and electrons in Boron, Sodium, and Phosphorus. Question 2 asks students to identify the diagram that best represents the nucleus of Beryllium from six choices. Question 3 requires students to fill in a table with the atomic number, mass number, number of protons, neutrons, and electrons for Aluminum, Hydrogen, Argon, and Magnesium using provided data.
Grade Level Suitability:
This worksheet is suitable for grades 7-9, specifically for students learning about atoms, elements, and the periodic table. The concepts of protons, neutrons, electrons, atomic number, and mass number are typically introduced in middle school science.
Why Use It:
This worksheet reinforces understanding of atomic structure, the periodic table, and how to determine the number of protons, neutrons, and electrons in an atom. It also helps students practice identifying elements and their properties.
How to Use It:
Students should use the provided periodic table and their knowledge of atomic structure to answer the questions. For question 1, they should look up the elements on the table and calculate the number of neutrons based on the atomic mass. For question 2, they need to understand what constitutes a nucleus. For question 3, they need to fill in the missing information using the provided data and the periodic table.
Target Users:
This worksheet is intended for middle school students studying chemistry or physical science. It's a good resource for homework, in-class practice, or review activities related to the periodic table and atomic structure.
This is a Periodic Table extra practice worksheet. It includes a simplified periodic table at the top. Question 1 asks students to determine the number of protons, neutrons, and electrons in Boron, Sodium, and Phosphorus. Question 2 asks students to identify the diagram that best represents the nucleus of Beryllium from six choices. Question 3 requires students to fill in a table with the atomic number, mass number, number of protons, neutrons, and electrons for Aluminum, Hydrogen, Argon, and Magnesium using provided data.
Grade Level Suitability:
This worksheet is suitable for grades 7-9, specifically for students learning about atoms, elements, and the periodic table. The concepts of protons, neutrons, electrons, atomic number, and mass number are typically introduced in middle school science.
Why Use It:
This worksheet reinforces understanding of atomic structure, the periodic table, and how to determine the number of protons, neutrons, and electrons in an atom. It also helps students practice identifying elements and their properties.
How to Use It:
Students should use the provided periodic table and their knowledge of atomic structure to answer the questions. For question 1, they should look up the elements on the table and calculate the number of neutrons based on the atomic mass. For question 2, they need to understand what constitutes a nucleus. For question 3, they need to fill in the missing information using the provided data and the periodic table.
Target Users:
This worksheet is intended for middle school students studying chemistry or physical science. It's a good resource for homework, in-class practice, or review activities related to the periodic table and atomic structure.




