0
Views
0
Downloads
0.0
0
Save
0 Likes
Ion and Charged Isotope Practice Worksheet
0 Views
0 Downloads
Paste this activity's link or code into your existing LMS (Google Classroom, Canvas, Teams, Schoology, Moodle, etc.).
Students can open and work on the activity right away, with no student login required.
You'll still be able to track student progress and results from your teacher account.
Information
Description
What It Is:
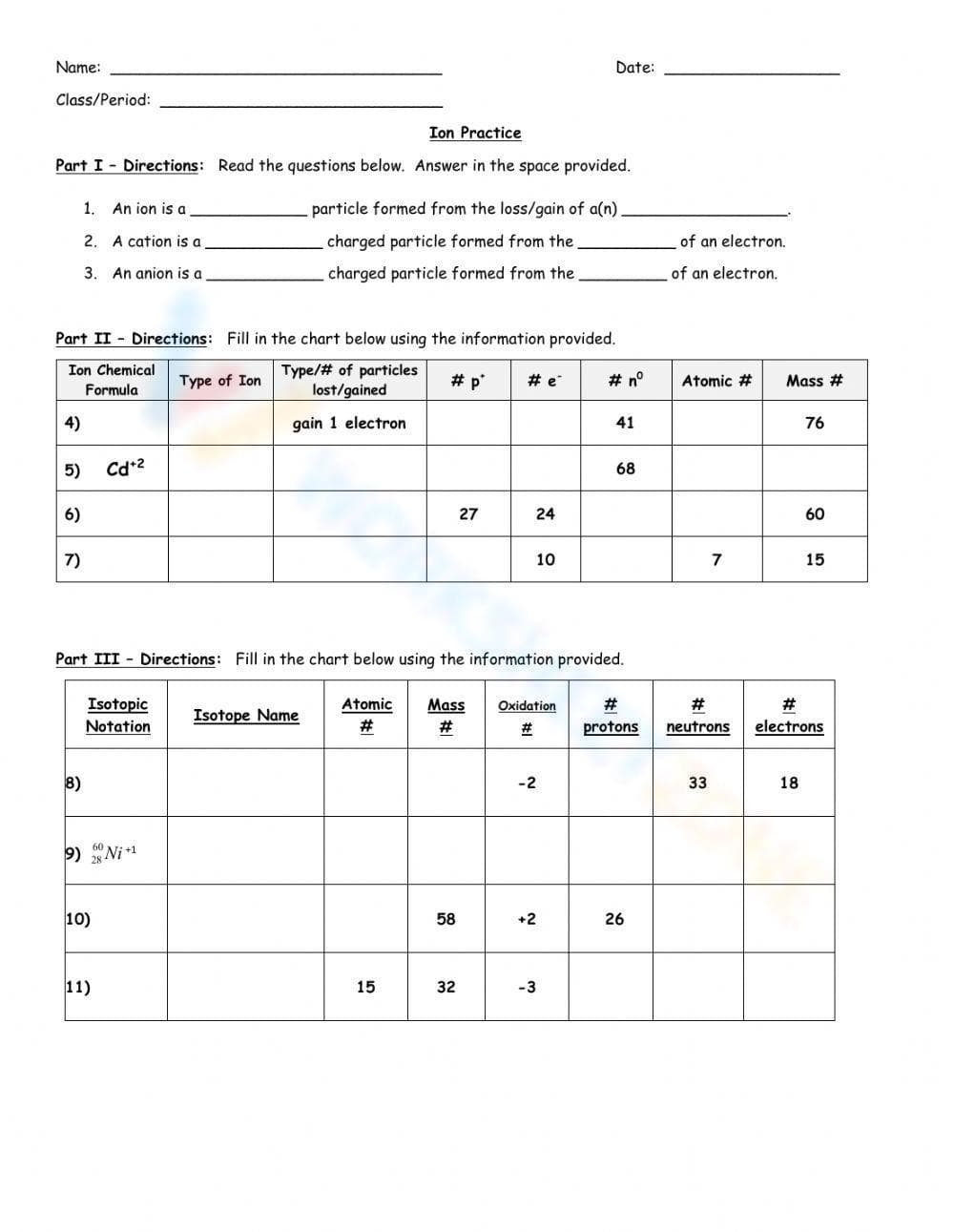
This is an 'Ion Practice' worksheet. It contains three parts. Part I includes fill-in-the-blank questions about the definition of an ion, a cation, and an anion. Part II requires students to fill in a table with information about ions, including the ion chemical formula, type of ion, number of particles lost/gained, number of protons, number of electrons, atomic number, and mass number. Part III requires students to fill in a table with information about isotopic notation, isotope name, atomic number, mass number, oxidation number, number of protons, number of neutrons, and number of electrons.
Grade Level Suitability:
This worksheet is suitable for high school chemistry students, specifically grades 9-12. It requires an understanding of atomic structure, ions, isotopes, and chemical formulas, which are typically covered in high school chemistry courses.
Why Use It:
This worksheet reinforces understanding of ions, cations, anions, isotopes, and their properties. It helps students practice calculating the number of protons, neutrons, and electrons in different ions and isotopes, and understand the relationship between atomic number, mass number, and charge.
How to Use It:
Students should read the directions carefully for each part. For Part I, they should fill in the blanks with the correct terms. For Parts II and III, they should use the provided information to complete the tables, calculating missing values based on their understanding of atomic structure and ion formation.
Target Users:
The target users are high school chemistry students who are learning about ions, isotopes, and atomic structure. It is appropriate for students who need extra practice or review on these concepts.
This is an 'Ion Practice' worksheet. It contains three parts. Part I includes fill-in-the-blank questions about the definition of an ion, a cation, and an anion. Part II requires students to fill in a table with information about ions, including the ion chemical formula, type of ion, number of particles lost/gained, number of protons, number of electrons, atomic number, and mass number. Part III requires students to fill in a table with information about isotopic notation, isotope name, atomic number, mass number, oxidation number, number of protons, number of neutrons, and number of electrons.
Grade Level Suitability:
This worksheet is suitable for high school chemistry students, specifically grades 9-12. It requires an understanding of atomic structure, ions, isotopes, and chemical formulas, which are typically covered in high school chemistry courses.
Why Use It:
This worksheet reinforces understanding of ions, cations, anions, isotopes, and their properties. It helps students practice calculating the number of protons, neutrons, and electrons in different ions and isotopes, and understand the relationship between atomic number, mass number, and charge.
How to Use It:
Students should read the directions carefully for each part. For Part I, they should fill in the blanks with the correct terms. For Parts II and III, they should use the provided information to complete the tables, calculating missing values based on their understanding of atomic structure and ion formation.
Target Users:
The target users are high school chemistry students who are learning about ions, isotopes, and atomic structure. It is appropriate for students who need extra practice or review on these concepts.




