1 / 2
0
Views
0
Downloads

0.0
0
Save
0 Likes
Honors Chemistry Worksheet
0 Views
0 Downloads
Paste this activity's link or code into your existing LMS (Google Classroom, Canvas, Teams, Schoology, Moodle, etc.).
Students can open and work on the activity right away, with no student login required.
You'll still be able to track student progress and results from your teacher account.
Information
Description
What It Is:
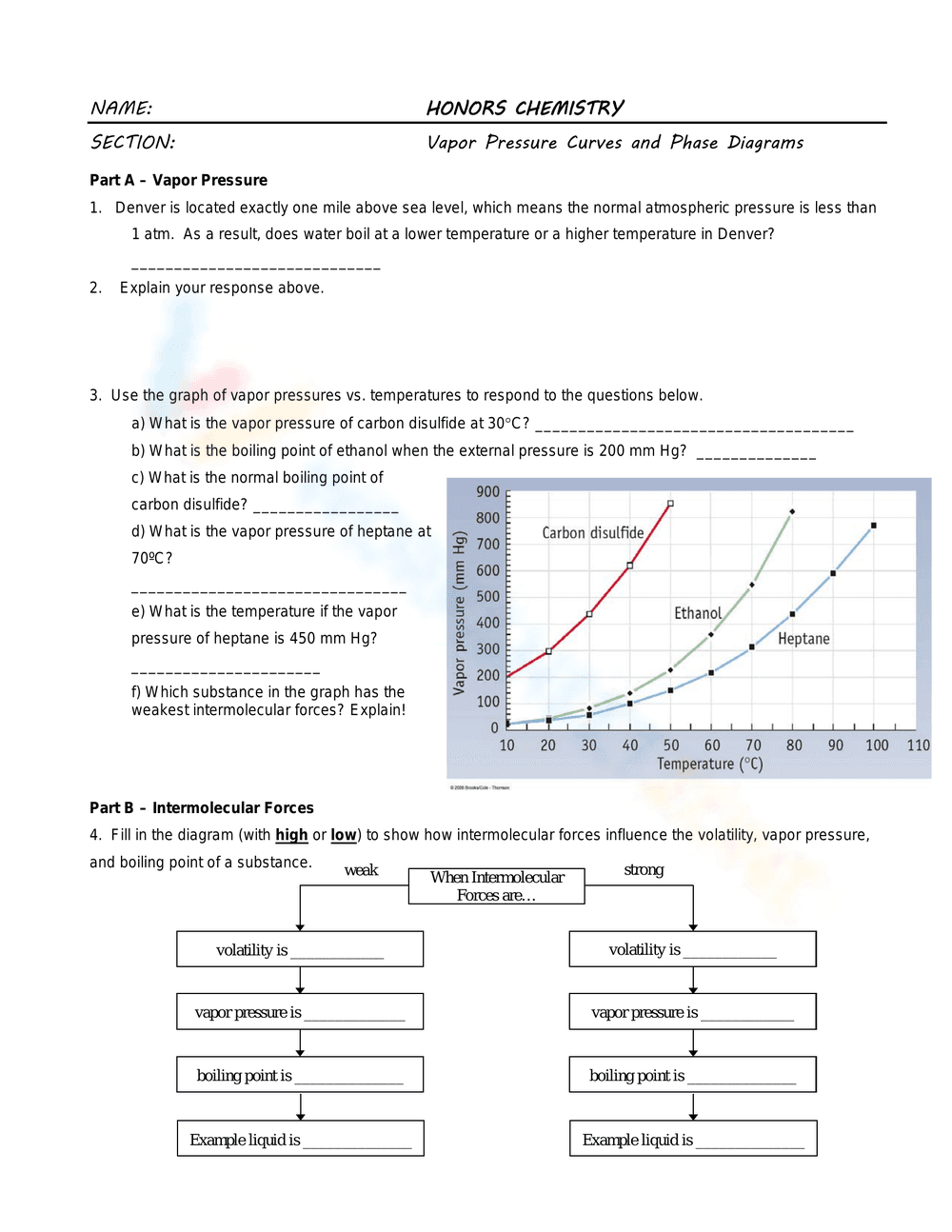
This is an Honors Chemistry worksheet focusing on vapor pressure curves, phase diagrams, and intermolecular forces. It includes questions about the effect of altitude on boiling point, using a graph to determine vapor pressures and boiling points of carbon disulfide, ethanol, and heptane at various temperatures and pressures. It also has a fill-in-the-blank diagram to illustrate how intermolecular forces affect volatility, vapor pressure, and boiling point.
Grade Level Suitability:
This worksheet is suitable for high school students, specifically grades 11-12, taking Honors Chemistry or AP Chemistry. The concepts and calculations require a solid understanding of chemistry principles.
Why Use It:
This worksheet helps students understand the relationship between vapor pressure, temperature, and boiling point. It reinforces the concepts of intermolecular forces and their impact on physical properties of substances. It also develops graph reading and data interpretation skills.
How to Use It:
Students should first read the introductory information and questions. They should then use the provided graph to answer questions related to vapor pressure and boiling points. Finally, they need to fill in the diagram with the appropriate terms (high or low) to show the relationship between intermolecular forces and physical properties.
Target Users:
This worksheet is designed for high school chemistry students, particularly those in Honors or AP Chemistry courses. It is also useful for teachers looking for supplemental materials to reinforce concepts related to vapor pressure, phase diagrams, and intermolecular forces.
This is an Honors Chemistry worksheet focusing on vapor pressure curves, phase diagrams, and intermolecular forces. It includes questions about the effect of altitude on boiling point, using a graph to determine vapor pressures and boiling points of carbon disulfide, ethanol, and heptane at various temperatures and pressures. It also has a fill-in-the-blank diagram to illustrate how intermolecular forces affect volatility, vapor pressure, and boiling point.
Grade Level Suitability:
This worksheet is suitable for high school students, specifically grades 11-12, taking Honors Chemistry or AP Chemistry. The concepts and calculations require a solid understanding of chemistry principles.
Why Use It:
This worksheet helps students understand the relationship between vapor pressure, temperature, and boiling point. It reinforces the concepts of intermolecular forces and their impact on physical properties of substances. It also develops graph reading and data interpretation skills.
How to Use It:
Students should first read the introductory information and questions. They should then use the provided graph to answer questions related to vapor pressure and boiling points. Finally, they need to fill in the diagram with the appropriate terms (high or low) to show the relationship between intermolecular forces and physical properties.
Target Users:
This worksheet is designed for high school chemistry students, particularly those in Honors or AP Chemistry courses. It is also useful for teachers looking for supplemental materials to reinforce concepts related to vapor pressure, phase diagrams, and intermolecular forces.




