0
Views
0
Downloads
0.0
0
Save
0 Likes
Calculating Atomicity
0 Views
0 Downloads
Paste this activity's link or code into your existing LMS (Google Classroom, Canvas, Teams, Schoology, Moodle, etc.).
Students can open and work on the activity right away, with no student login required.
You'll still be able to track student progress and results from your teacher account.
Information
Description
What It Is:
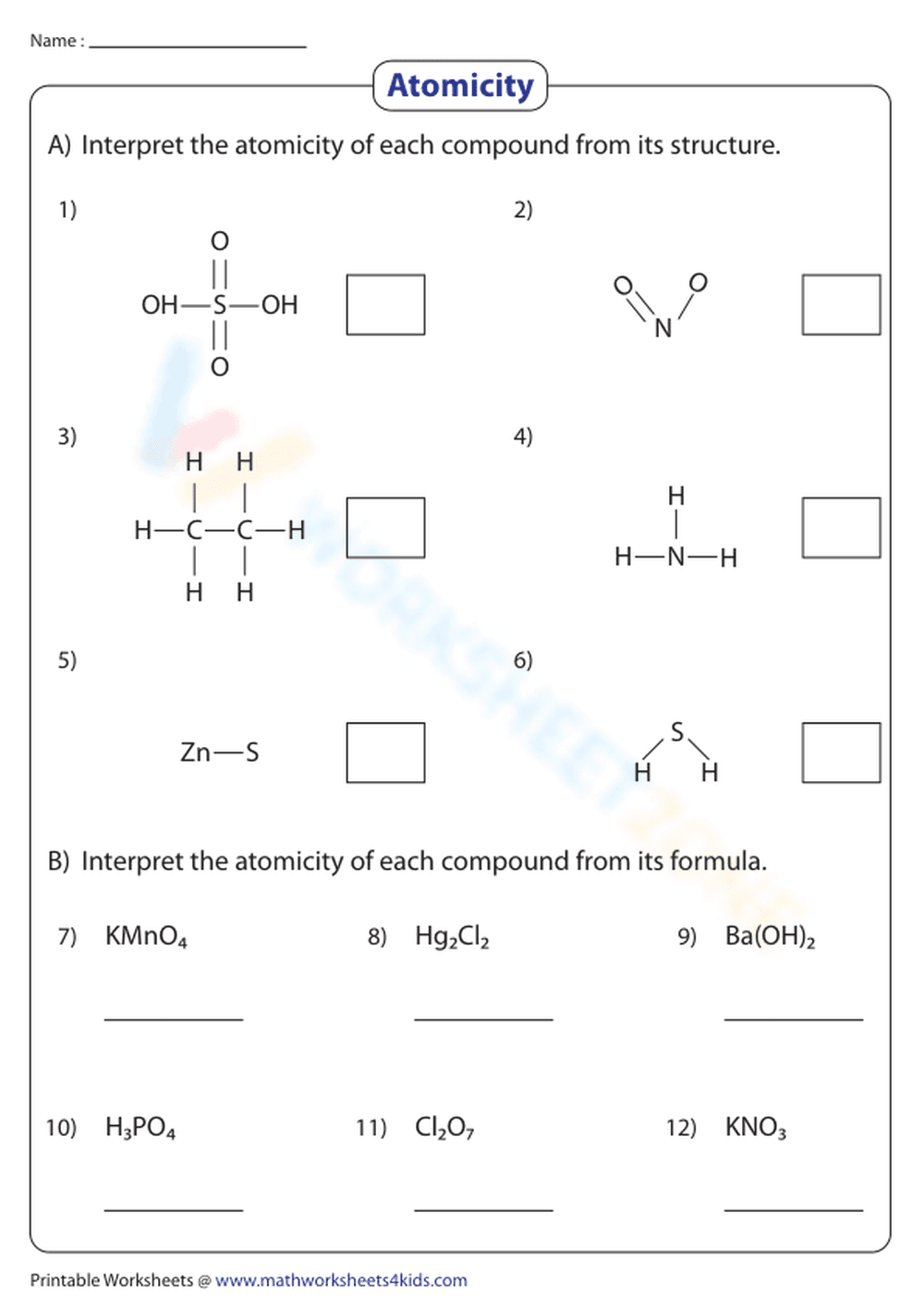
This is a chemistry worksheet focusing on atomicity. The worksheet is divided into two sections. Section A presents structural formulas of compounds (e.g., H2SO4, N2O, C2H6, NH3, ZnS, H2S) and requires the student to interpret the atomicity from the structure. Section B provides chemical formulas (e.g., KMnO4, Hg2Cl2, Ba(OH)2, H3PO4, Cl2O7, KNO3) and asks the student to determine the atomicity based on the formula. There are blank spaces provided for answers next to each compound.
Grade Level Suitability:
This worksheet is suitable for grades 9-12, specifically for high school chemistry students. The concepts of atomicity and chemical formulas are typically introduced in high school chemistry courses. It could also be used as a review for introductory college chemistry.
Why Use It:
This worksheet helps students understand the relationship between chemical structure, chemical formula, and atomicity. It reinforces their understanding of chemical nomenclature and the ability to interpret chemical formulas and structural diagrams. It also helps develop critical thinking skills in determining the number of atoms of each element in a compound.
How to Use It:
Students should first understand the definition of atomicity. Then, for Section A, students need to count the number of atoms of each element present in the structural formula and write the total number of atoms for each element in the provided blank space. For Section B, students need to count the number of atoms of each element present in the chemical formula and write the total number of atoms for each element in the provided blank space.
Target Users:
The target users are high school chemistry students, teachers looking for supplementary material, and students reviewing chemistry concepts. It can also be used by tutors and homeschooling parents.
This is a chemistry worksheet focusing on atomicity. The worksheet is divided into two sections. Section A presents structural formulas of compounds (e.g., H2SO4, N2O, C2H6, NH3, ZnS, H2S) and requires the student to interpret the atomicity from the structure. Section B provides chemical formulas (e.g., KMnO4, Hg2Cl2, Ba(OH)2, H3PO4, Cl2O7, KNO3) and asks the student to determine the atomicity based on the formula. There are blank spaces provided for answers next to each compound.
Grade Level Suitability:
This worksheet is suitable for grades 9-12, specifically for high school chemistry students. The concepts of atomicity and chemical formulas are typically introduced in high school chemistry courses. It could also be used as a review for introductory college chemistry.
Why Use It:
This worksheet helps students understand the relationship between chemical structure, chemical formula, and atomicity. It reinforces their understanding of chemical nomenclature and the ability to interpret chemical formulas and structural diagrams. It also helps develop critical thinking skills in determining the number of atoms of each element in a compound.
How to Use It:
Students should first understand the definition of atomicity. Then, for Section A, students need to count the number of atoms of each element present in the structural formula and write the total number of atoms for each element in the provided blank space. For Section B, students need to count the number of atoms of each element present in the chemical formula and write the total number of atoms for each element in the provided blank space.
Target Users:
The target users are high school chemistry students, teachers looking for supplementary material, and students reviewing chemistry concepts. It can also be used by tutors and homeschooling parents.




